January 2, 2024
Voluntary Recall: Nutramigen with Probiotic LGG Powder Infant Formula
Mead Johnson has announced a voluntary recall of select batches of POWDER Nutramigen with Probiotic LGG Powder Infant Formula in 12.6 and 19.8 oz. cans due to a possibility of contamination. NO illness or adverse consumer reactions have been reported to date.
NO Nutramigen liquid formulas (concentrate and ready to feed) products are impacted.
Details: This recall impacts Nutramigen with Probiotic LGG Powder Infant Formula in 12.6 and 19.8 oz. cans manufactured in June 2023 and distributed primarily in June, July, and August 2023. The products have a UPC Code of 300871239418 or 300871239456 and “Use By Date” of “1 Jan 2025”. The batches in question can be identified by the batch code on the bottom of the can.
The following recalled product batch codes and can size associated with each batch were distributed in the U.S.:
• ZL3FHG (12.6 oz cans)
• ZL3FMH (12.6 oz cans)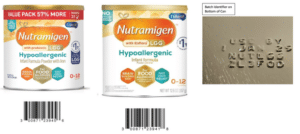
• ZL3FQD (12.6 oz cans)
• ZL3FRW (19.8 oz cans)
• ZL3FXJ (12.6 oz cans)
Recall link:
What you should do if you are using this product: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/reckittmead-johnson-nutrition-voluntarily-recalls-select-batches-nutramigen-hypoallergenic-infant
1. Check the bottom of each can to identify whether the batch number is affected.
2. If you do have some with these batch numbers, return it to your local WIC office.
3. WIC staff will assist with replacing eligible current benefits.
4. Parents and caregivers of infants who have used this product and are concerned about the health of their child should contact their health care provider.
Need help? Call the Adair County Health Department at (660) 665-8491.
